We have all experienced that frustrating moment when a trusty steam iron starts to fail. Perhaps it is a sudden puff of white powder on a navy blazer, or maybe the steam vents that used to hiss with power are now barely whispering. When an iron dies, we often blame the brand or the age of the device. However, the most common culprit is actually flowing right out of your kitchen faucet.
Hard water is the silent assassin of small household appliances, and steam irons are particularly vulnerable. To understand why your iron is struggling, we need to dive into the chemistry of water and look at the microscopic processes happening inside your appliance every time you plug it in.
By the way, has limescale destroyed your iron? Here are the best steam irons to buy today.
What Exactly Is Hard Water?
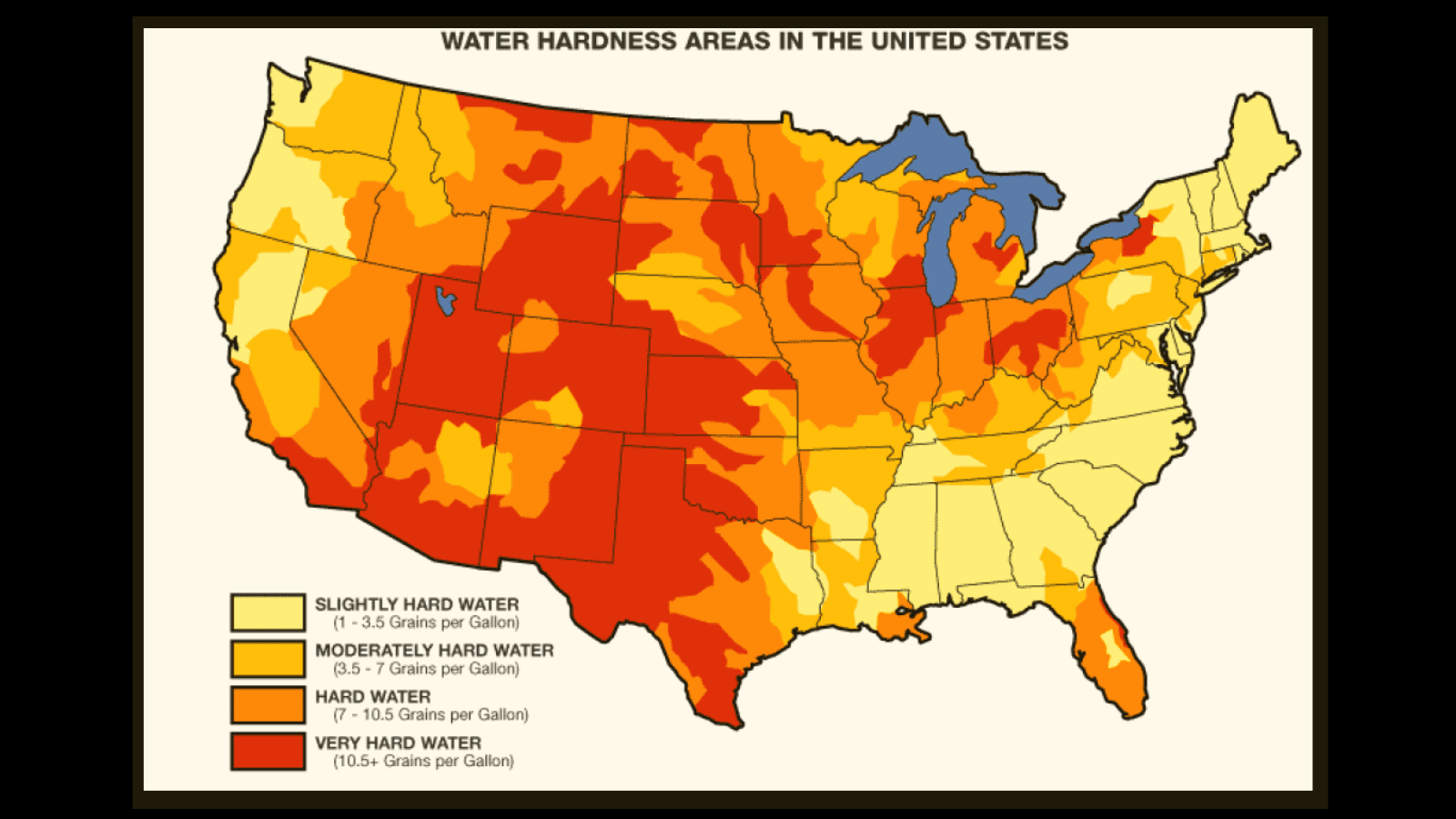
When we talk about water being “hard,” we are not talking about its physical texture. Hardness refers to the concentration of dissolved minerals within the water. As rainwater falls, it is naturally soft. However, as it filters through the ground and into our water systems, it picks up minerals from rocks and soil.
The primary minerals involved in water hardness are calcium and magnesium. Specifically, these minerals exist in the form of calcium cations and magnesium cations. In many regions, water also contains bicarbonates. When these elements meet, they create a solution that is perfectly clear to the eye but chemically “heavy.”
The degree of hardness is usually measured in grains per gallon (GPG) or parts per million (PPM). If your home has more than 120 PPM of these minerals, you are officially in the hard water zone. While these minerals are perfectly safe and even healthy for human consumption, they are absolute poison for the internal heating elements of a steam iron.
The Chemical Transformation: From Liquid to Limescale
The reason hard water kills irons so effectively lies in a simple chemical reaction triggered by heat. Inside your iron is a steam chamber. When you fill the tank and turn the dial to a high setting, the heating element warms the metal walls of this chamber to temperatures well above the boiling point of water.
When the liquid water touches that hot surface, it undergoes a phase change into steam. However, minerals like calcium and magnesium cannot turn into gas. They are left behind. As the water evaporates, the concentration of these minerals in the remaining droplets increases until they can no longer stay dissolved.
This leads to the formation of solid calcium carbonate. This is the white, chalky substance we know as limescale. It starts as a microscopic film, but with every shirt you iron, that film grows thicker and harder.
How Limescale Destroys an Iron from the Inside Out
Limescale does not just sit there quietly. It actively interferes with the mechanical and thermal processes that make your iron work. Here is a breakdown of the three main ways limescale acts as an iron killer.
1. The Insulator Effect
Calcium carbonate is a very poor conductor of heat. In fact, it is often used as an industrial insulator. When a layer of scale builds up on the heating element or the walls of the steam chamber, it creates a barrier between the heat source and the water.
Your iron’s thermostat senses that the water is not turning into steam fast enough, so it keeps the heating element turned on for longer. This causes the internal components to overheat, leading to a shorter lifespan for the motor and the electrical connections. You might notice your iron feels much hotter to the touch on the handle or the casing, even though the steam output is weak.
2. Blocked Vents and “Spitting”
As the scale flakes off the internal walls, the debris has to go somewhere. It naturally migrates toward the smallest openings: the steam vents on the soleplate. As these holes become partially blocked, the pressure inside the iron increases.
Instead of a fine mist, the iron begins to “spit” hot water. This happens because the steam cannot escape fast enough, and the pressure forces unvaporized water out through the gaps. If that water has been sitting in a scaly chamber, it will carry brown or white sediment with it, potentially ruining your clothing.
3. Corrosion and Seal Degradation
Limescale is porous. It can trap moisture against the metal parts of the iron even when the tank is empty. This prolonged exposure to moisture, combined with the alkaline nature of the minerals, can lead to corrosion. Over time, this corrosion eats away at the rubber seals and gaskets that keep the water tank separate from the electrical housing. Once those seals fail, the iron will begin to leak from the base or the handle, which is often a terminal issue for the device.
Why Your Steam Iron Is More Vulnerable Than Other Appliances
You might wonder why your kettle or dishwasher seems to handle hard water better than your iron. The answer is the size of the plumbing. A dishwasher has large pipes and high volume water flow that can flush out some sediment. A steam iron, however, relies on capillary action and tiny, precision engineered channels to move water.
In a modern steam iron, some of the internal tubes are barely wider than a needle. It takes very little mineral buildup to create a total blockage in a system this small. Furthermore, the “flash boiling” process used in irons is a more aggressive way to create scale than the slower heating process used in a traditional water heater.
Identifying the Signs of Limescale Damage
Limescale is a progressive problem. If you catch it early, you can often reverse the damage. If you wait too long, the “clog” becomes a solid mineral brick that no amount of cleaning can fix. Watch for these four warning signs:
- The White Dust: If you see white, powdery flakes on your ironing board, the scale is already brittle and breaking off inside the tank.
- Delayed Steaming: If you press the steam trigger and there is a three or four second delay before anything happens, the internal pump is struggling against a mineral blockage.
- The “Drag” Factor: If the soleplate feels sticky or has a visible white film, the minerals are beginning to coat the outside of the iron.
- Strange Noises: A loud humming or vibrating sound from the pump often means it is trying to push water through a narrowed, scaly pipe.
How to Fight Back: The Science of Descaling
Since we cannot change the chemistry of the water coming out of the ground easily, we have to change how we maintain our tools. Descaling is the process of using an acid to dissolve the alkaline calcium carbonate.
Common acids used for this include citric acid (found in lemon juice) or acetic acid (found in white vinegar). When these acids touch limescale, a chemical reaction occurs that turns the solid calcium carbonate back into a soluble liquid that can be flushed away.
However, as we have noted in previous guides, you must be careful. While vinegar is great at eating scale, it can also be hard on the rubber seals inside high-end irons. This is why many manufacturers recommend a dedicated descaling solution or a simple “thermal shock” method using the built-in self-clean button.
Prevention: The Best Way to Extend Your Iron’s Life
If you want your iron to last a decade instead of two years, you have to manage the mineral input.
The 50/50 Solution
For most people, using a mix of half tap water and half distilled water is the perfect balance. The distilled water reduces the total mineral count, while the tap water provides enough conductivity for the iron’s internal sensors to work correctly.
Emptying the Tank
Always empty the water reservoir after every use. When water sits in the tank, it evaporates slowly, leaving a higher and higher concentration of minerals behind. By emptying the tank, you ensure that the internal chamber stays dry and scale-free between sessions.
The “Bottom Up” Clean
Wipe the soleplate with a damp cloth after every use. This prevents minerals from hardening on the outside of the vents, which is often where the most stubborn blockages begin.
Protecting Your Steam Iron
Limescale might seem like a minor annoyance, but on a molecular level, it is a formidable opponent for your household appliances. By understanding that heat plus hard water equals solid rock, you can see why maintenance is not optional.
Protecting your iron from the “number one killer” is as simple as being mindful of the water you use and taking five minutes to flush the system once a month. Your clothes, your wallet, and your iron will thank you for it.
